Technologies
Symic Bio Improving Vascular Interventions with a Novel Drug –
An Interview with John Paderi of Symic Bio
What is the need Symic Bio seeks to address?
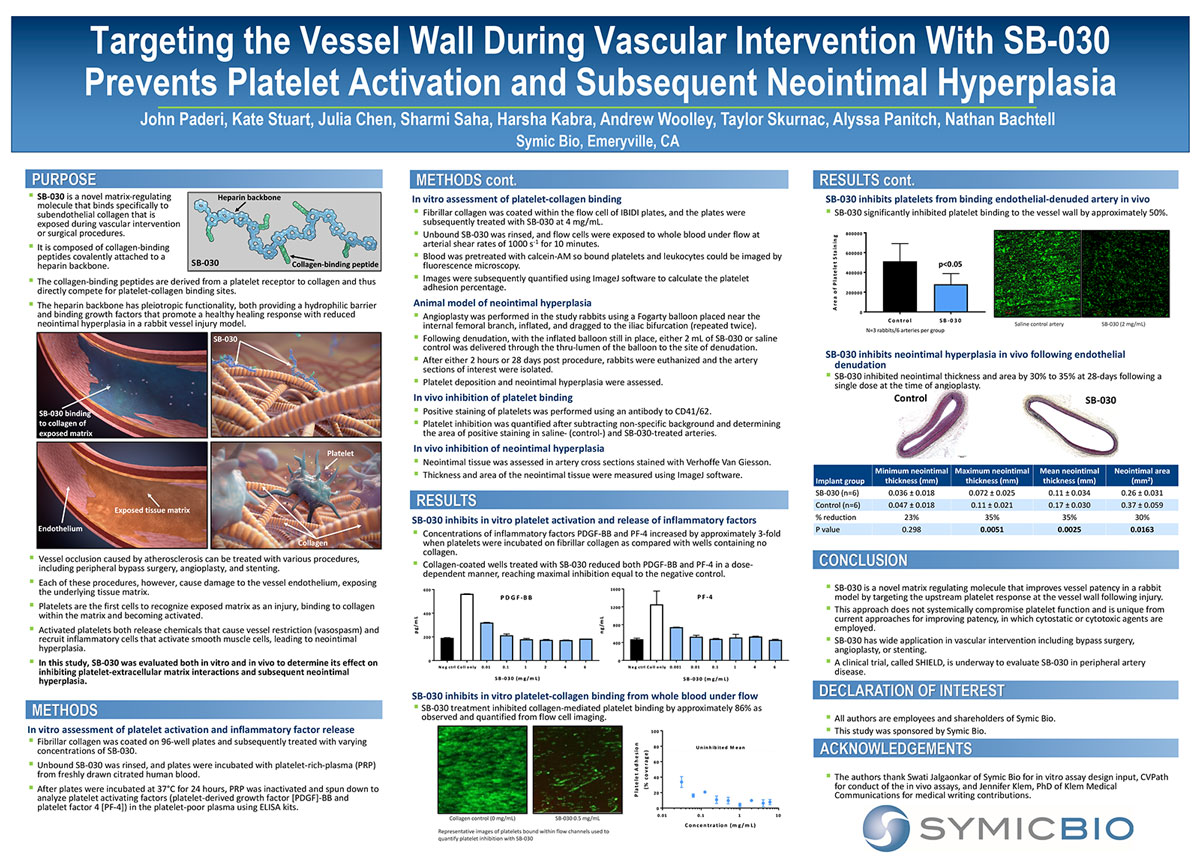
When people develop atherosclerosis, a peripheral occlusion from the build-up of plaque in a blood vessel, interventions such as peripheral bypass surgery, angioplasty, and stenting can be used to open the vessel and restore blood flow. Often, however, these procedures inadvertently damage the endothelium, the interior lining of the vessel, exposing the tissue matrix beneath. The matrix contains collagen, which attracts platelets, tiny blood cells that help your body form clots. The platelets rush to the site of injury, bind to the matrix, and activate to help fix the damage. The problem with this healing response is that the activated platelets have multiple adverse effects, including attracting inflammatory cells to the injury and leading to scarring that can create new occlusions. The new occlusions can be much more difficult to treat and, depending on the procedure or location of the vessel, can lead to amputation or death.
“We wondered if we could do something upstream, as part of the preliminary procedure, to improve clinical outcomes for these patients….”
While current drug-eluting and interventional technologies have led to improvements in this field, challenges remain, including long-term antiplatelet use and, more recently, concern about the systemic impact of the drugs used in these technologies and their impact on amputation and overall patient survival. We wondered if we could do something upstream, as part of the preliminary procedure, to improve clinical outcomes for these patients and offer a safer, more robust treatment approach to vascular interventions.
What key insight was most important to guiding the design of your solution?
Through our research, we realized that the role of platelets was somewhat overlooked in these procedures. They were recognized for contributing to blood clots, but less appreciated for the part they play in inflammation and scarring inside the blood vessel. We realized that if we could come up with a way to locally prevent platelets from binding to the collagen in the exposed tissue matrix, then we could inhibit upstream adverse effects. So we went deep into the research in order to understand platelet-collagen interactions and how we could potentially affect them.
 Members of the Symic Bio team, including John Paderi (second from left), celebrating the close of their series A funding.
Members of the Symic Bio team, including John Paderi (second from left), celebrating the close of their series A funding.
How does your solution work?
Our solution, currently called SB-030, is a peptide drug that is conjugated to heparin. This drug competitively inhibits local platelet activation at the injured vessel wall. In our clinical trials to date, SB-030 is delivered locally to the vessel during a procedure. By binding to the injured vessel wall, SB-030 prevents platelets from binding and activating and, thus, the downstream adverse effects are avoided. In this way, the vessel remains open without scarring. The value of SB-030, however, is that it offers a safer, more robust and versatile approach compared to the current drug-eluting or coated technologies.
 This short promotional video, provided by Symic Bio, illustrates how the company's drug is intended to work (see endorsement disclaimer below).
This short promotional video, provided by Symic Bio, illustrates how the company's drug is intended to work (see endorsement disclaimer below).
At what stage of development is the solution?
SB-030 is moving into phase II clinical trials on its way toward patient care.
Tell us about a major obstacle you encountered and how you overcame it.
Given that the field of vascular intervention has been around for a while, including the development of drugs used in combination with various devices, some stakeholders are of the opinion that the issues are solved and new innovation is not needed. This varies depending on the part of the body that is affected and, in general, there is not a particularly strong consensus among interventionalists on whether new approaches are needed. Rather than working to convince the whole field of the need and opportunity, we started exploring vascular interventions where the pathology is the same but the need is more apparent. This led us to peripheral vein grafts, where the failure rates are abysmally high and there’s more widespread demand for new solutions. Making the pivot to an area with a recognized gap in current therapies was pretty critical for us. It helped us raise money and get partners interested. Following the clinical need was particularly interesting with respect to the FDA, as they encouraged us to move more rapidly and skip over a phase II trial and go directly into phase III.
“Making the pivot to an area with a recognized gap in current therapies was pretty critical for us.”
What advice do you have for other innovators about health technology innovation?
I think the most important advice is to build and cultivate a great team. There are inevitably numerous setbacks and roadblocks that come up along the way. The best way to prepare is to make sure you have a dedicated and passionate team, because it's that team that's going to navigate the obstacles. And if nothing else, it's that team that's going to make you happy waking up every day and working together to overcome those challenges.
Another bit of advice I learned along the way is that it’s important to have a clear, compelling story. Our solution is a molecule that has multiple mechanisms. For example, in addition to affecting platelet-collagen interactions, SB-030 has anti-fibrotic properties, which is quite beneficial for this use in vascular intervention. While it might appear this would be a benefit, it can actually be confusing to partners and investors. In particular, in the pharma world, there is a general view that if your molecule does more than one good thing, it may also do a bunch of bad things. So, over time, we recognized the need to tell one crisp story that sticks.
John Paderi completed the Biodesign Fellowship in 2013. He co-founded Symic Bio in 2012 and served as the company’s Vice President of Operations until 2019. To learn more, visit the Symic Bio website.
Disclaimer of Endorsement: All references to specific products, companies, or services, including links to external sites, are for educational purposes only and do not constitute or imply an endorsement by the Byers Center for Biodesign or Stanford University.