Technologies
Zenflow A Less Invasive Solution for Benign Prostatic Hyperplasia:
An Interview with Nick Damiano of Zenflow
As a Biodesign Fellow, what was the need you set out to address?
The clinical focus in 2013-14 was urology and nephrology. We went into the urology clinics and saw that almost all of the men who were there had some kind of issue with benign prostatic hyperplasia (BPH). Even if that wasn’t the primary reason for their visit, they were complaining about it, and they didn’t like the existing solutions. The market has changed a lot since then but, at the time, the only choices were drugs, which aren’t always effective and can have unpleasant side effects, or TURP, a highly-invasive surgery with a significant risk of complications. We wanted to develop a minimally-invasive approach with a lower risk profile.
Since then, other minimally-invasive solutions have become available, including the UroLift device by NeoTract, which was one of Josh Makower’s companies [Josh is co-founder of Stanford Biodesign]. That gave us pause, but we felt that we had an idea that could be better for the patient and the urologist. That’s the premise we started with, and we’ve only become more confident in our approach since then.
What key insight was most important to guiding the design of your solution?
“It was patient perspectives that led us down the path to our current device.”
It was patient perspectives that led us down the path to our current device. We learned that they really cared about the ease of the procedure, not having a rigid scope put in, the length of the recovery time, not needing a catheter after treatment. Those kinds of things were really important.
How does your solution work?
The Spring device is a small nitinol implant that’s deployed through a flexible cystoscope in the urologist’s office. The implant design was driven by extensive studies of the anatomy and disease state and lots of iteration on the bench. The delivery system mechanics have become complicated since it wasn’t easy to make the deployment accurate and reliable. But on the urologist’s end, it’s a trigger-based mechanism that is intended to be easy to learn and execute. The end goal is to provide a treatment that’s less painful for the patient – both during the procedure and during recovery; safe and effective in over the long term; and as simple as possible for the urologist to use. While it wasn’t easy to develop a product to meet all of these needs, we feel good about where we are now.
 Zenflow co-founders Shreya Mehta and Nick Damiano.
Zenflow co-founders Shreya Mehta and Nick Damiano.
At what stage of development is the solution?
We'v collected all of the clinical data from our first-in-human studyusing the first-generation device. Our theory is that this approach can be less traumatic and lead to faster recovery, which we are evaluating in this series. We have followed all of the patients out to 12 months and will be publishing both short and long-term data in the near future.
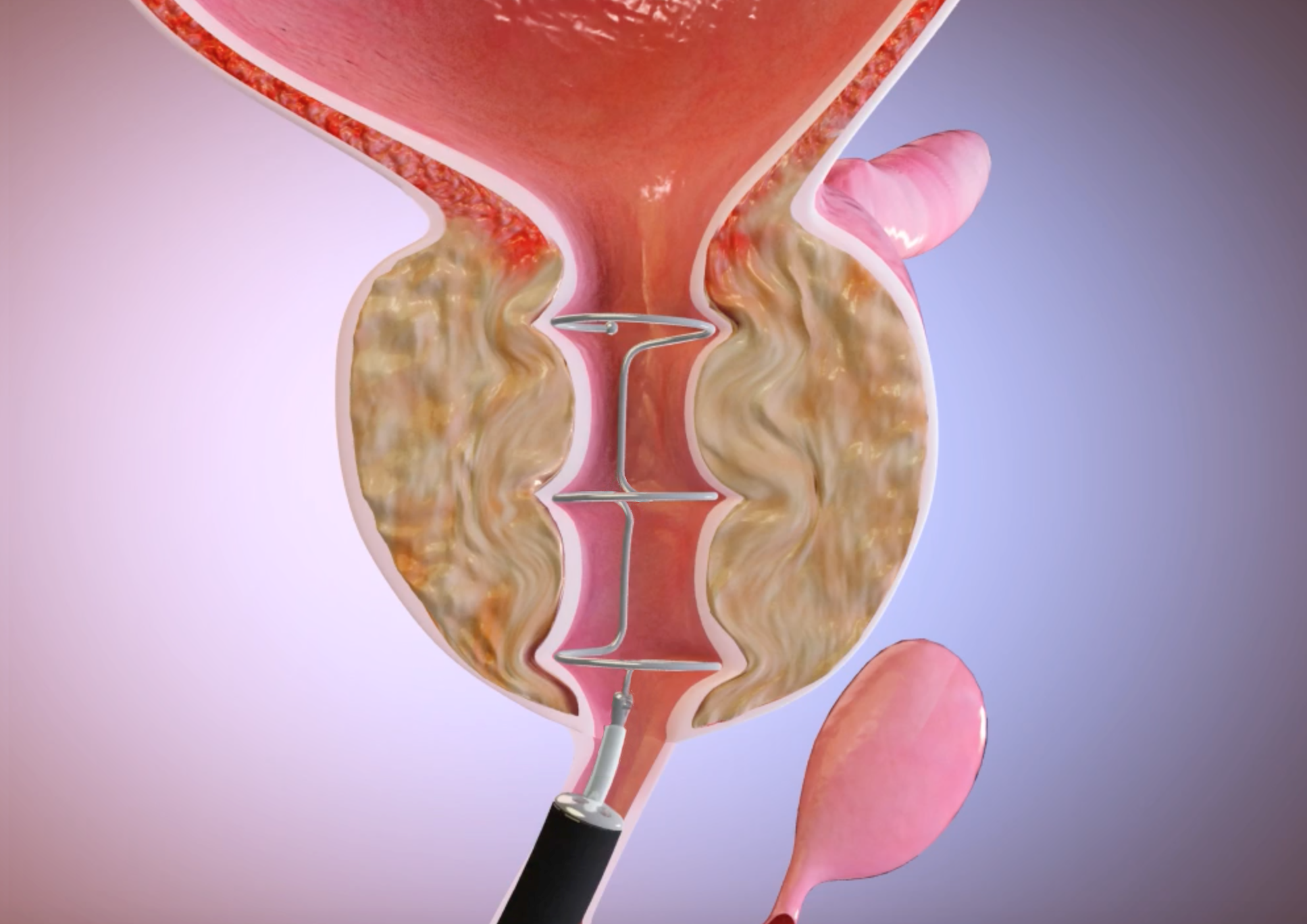 The Zenflow Spring device is a small nitinol implant deployed through a flexible cystoscope to provide a less invasive treatment for benign prostatic hyperplasia.
The Zenflow Spring device is a small nitinol implant deployed through a flexible cystoscope to provide a less invasive treatment for benign prostatic hyperplasia.
We’ve recently developed a second-generation product that we hope to use for early commercialization. In 2018, we plan to start larger studies in Europe and the US aimed at securing regulatory approvals and giving urologists the data they need to begin using the device in their everyday practice. We’re now closing a Series A round that will support all of this work. It’s going to be an exciting next couple of years.
Tell us about a major obstacle you encountered and how you overcame it.
Our initial clinical trials were pretty tricky to navigate. With the first cases, we had some issues deploying the device – it was tougher than expected to get into place. Fortunately, we almost always ended up positioning it appropriately, and once we got it in place it worked really well. But the difficulty of deployment, which prevented us from gaining sufficient confidence to perform the procedure in the office under local anesthesia, made urologists more hesitant to enroll patients than they otherwise would have been.
This created a problem because we only had three investigational sites. One urologist was hesitant to enroll patients because of the difficult deployment and inability to do the procedure in the office. Another was traveling frequently and didn’t have time to identify, screen, and treat patients. The third was based in a small town and only had a small potential patient pool to draw from. Ultimately, we ended up traveling to two of our sites in New Zealand and spent three weeks working with them to identify potential patients. We finally got one more patient, and then a few more after that, and eventually completed our trial enrollment.
This experience threatened our finances as well, because you really need patient data to raise significant commercial funding. When your sites are not enrolling, it gets stressful – especially while you continue to burn cash.
Reflecting on your experience, what advice do you have for other health technology innovators?
One key insight I would share is that most people misunderstand why therapies that failed in the past didn’t work. Really understanding this can be key to developing something that does work. This realization led to many more insights for the Zenflow team. We’ve made a point to always continue learning more about the disease state, anatomy, and urologists’ prior experience with BPH therapies, which has been invaluable as we’ve continued to improve our device.
“Most people misunderstand why therapies that failed in the past didn’t work…. [this] led to many more insights for the Zenflow team.
I would also advise other innovators not to underestimate the challenges involved in launching a medtech start-up, or to think they're going to be an exception to the rule. It's really difficult. But, then again, it’s definitely not impossible!
Nick Damiano co-founded Zenflow from the 2013-14 Biodesign Innovation Fellowship with fellowship alumna Shreya Mehta. To learn more, visit the Zenflow website.
Disclaimer of Endorsement: All references to specific products, companies, or services, including links to external sites, are for educational purposes only and do not constitute or imply an endorsement by the Byers Center for Biodesign or Stanford University.



