Technologies
Prescient Surgical Reducing the Risk of Surgical Infections:
An Interview with Jon Coe, Jeremy Koehler, and Insoo Suh of Prescient Surgical
As Biodesign Innovation Fellows, what is the need you set out to address?
Insoo: Even though we come from different backgrounds, we were all interested in advancing surgical practice. During our clinical observations, one problem that kept coming up was surgical site infections [SSIs]—infections that develop after a procedure at the incision site. One day, we watched a kidney transplant procedure in which the attending physician considered infection to be an inevitable outcome. That experience brought the need into sharp focus. Surgeons have an innate sense for which patients have the highest risk of contracting an SSI. So why weren’t they able to proactively intervene to mitigate that possibility? That realization sparked our efforts to find a way to address the root causes of infection during surgery.
“Surgeons have an innate sense for which patients have the highest risk of contracting an SSI. So why weren’t they able to proactively intervene to mitigate that possibility?”
Jon: At the time we were Biodesign Innovation Fellows, SSI reduction recently had been identified as a national priority as part of an overall effort to reduce infections acquired by patients in healthcare facilities. Medicare rules require hospitals to report their rate of hospital-acquired infections, which include SSI, to the Centers for Disease Control and Prevention’s National Healthcare Safety Network. Poor performing hospitals risk losing 1% of their Medicare revenue every year. In light of these policy changes, we realized that developing a technology to prevent SSIs would not only help patients, but could potentially reduce healthcare costs.
What key insight was most important in guiding the design of your solution?
Insoo: First, we established a deep understanding of the pathophysiology of surgical site infection. Then we studied existing surgical practices and current methods of infection control. There were a number of problems. Incomplete skin antisepsis introduces bacteria into the incision, and passive wound protectors can’t clear the invading bacteria. Manual irrigation is disruptive and can spread bacteria. And prophylactic antibiotic concentrations at the incision can fall below inhibitory levels.
“We wanted to design an approach that would concentrate the therapy where the threat of bacterial contamination is most critical.”
Jeremy: SSIs are a local phenomenon, so our insight was to target the delivery of the therapy at the surgical site. In contrast to systemic therapies that are circulated throughout the body to achieve a preventative effect at the surgical site, we wanted to design an approach that would concentrate the therapy where the threat of bacterial contamination is the most critical. We wanted to develop a device that a surgeon could use during the procedure that would diffuse a therapeutic fluid directly into the tissue to actively cleanse the incision site.
How does your solution work?
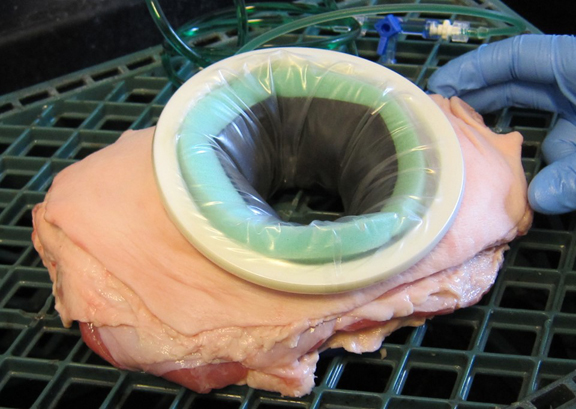
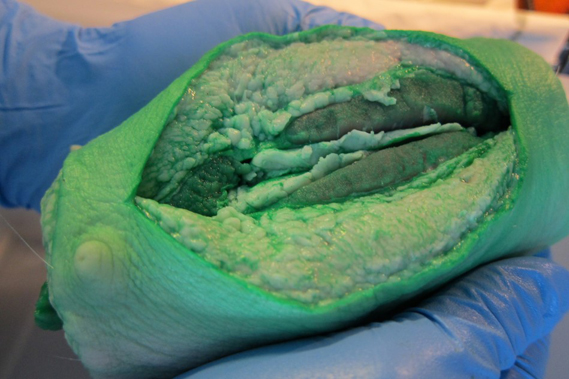
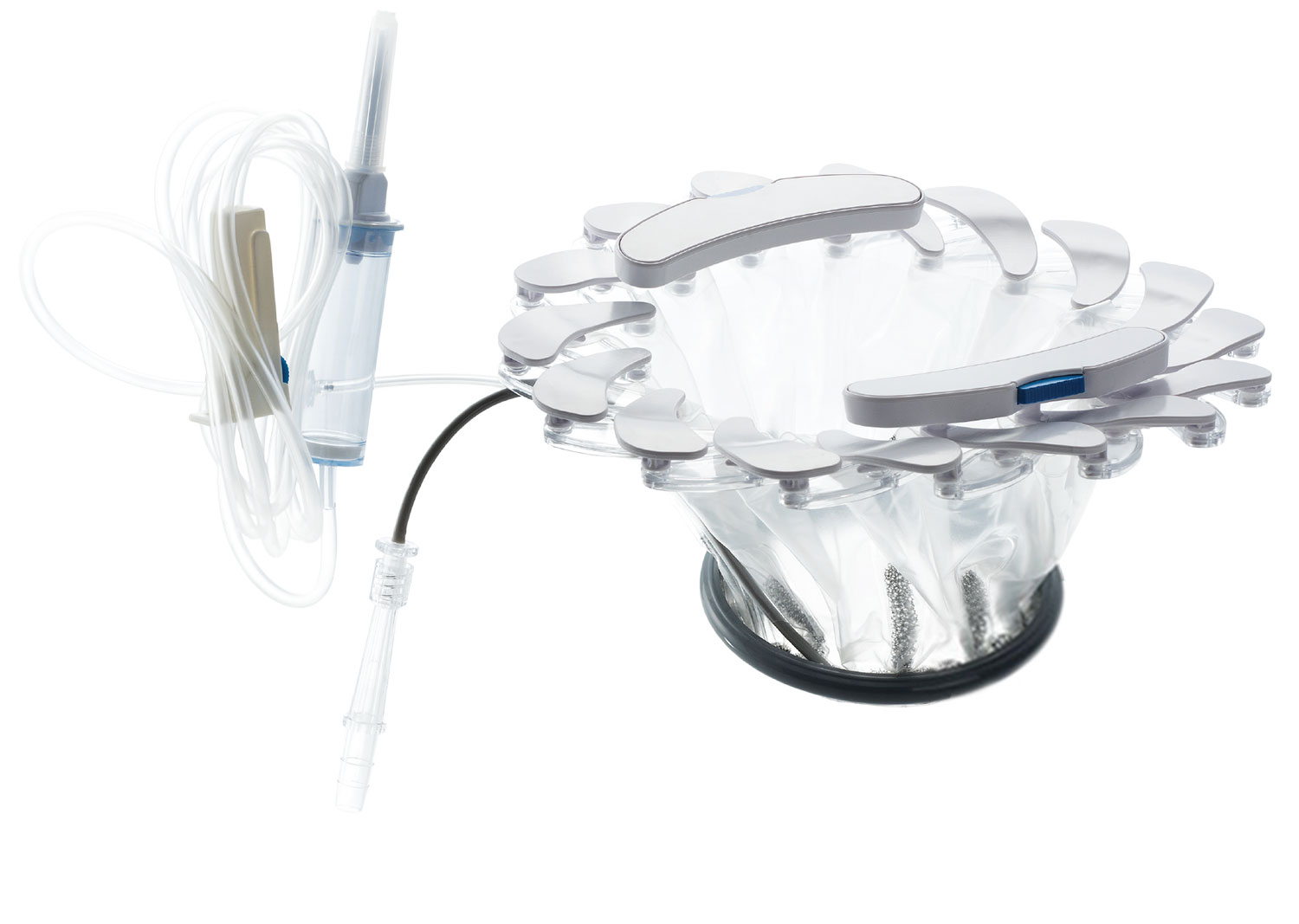
Jon: Our solution, called CleanCision, is a first-in-class technology that continuously clears harmful bacteria from the incision, combining wound protection and irrigation into an intuitive and easy-to-use retraction system.
Jeremy: The device provides access to the surgical site and uses a sterile irrigant solution to clear contamination and prevent it from invading the surgical incision. The wound edge is continuously irrigated while suction removes contaminants throughout the surgery.
At what stage of development is the solution?
Jon: CleanCision is currently commercially available in the US and Europe and we’re working directly with leading hospital systems to integrate it into their infection control protocols. Our initial focus is on abdominal surgery and particularly colorectal surgery, where the risk, frequency, and severity of surgical site infection is high and the need is acute.
Insoo: Now that the product is in use, we’re seeing that a proactive approach to clearing contamination during surgery has the potential to better protect patients from infection and help hospitals address the increased health care costs that result from surgical site infections, such as extended hospital stays, re-hospitalization, and rising infection rates that trigger penalties from CMS.
 This short promotional video, provided by Prescient Surgical, illustrates how the CleanCision technology is intended to work (see endorsement disclaimer below).
This short promotional video, provided by Prescient Surgical, illustrates how the CleanCision technology is intended to work (see endorsement disclaimer below).
What are your plans for the future?
Jon: In parallel with driving increased adoption of CleanCision, we’re focused on expanding into other surgical areas and working on new technologies to make surgery safer and improve post-operative outcomes.
Tell us about an obstacle you encountered and how you overcame it?
Jeremy: Although our concept was intuitive, developing a design that would hold the wound open and deliver fluid to the tissue proved challenging. For weeks, Jon and I worked nights in the Stanford Biodesign prototyping lab, running through more than a dozen different prototypes including an inflatable model, one with pull cords, and another that used a spiral mechanism. We were cranking through a prototype a week, just racking our brains and testing the limits of the fabrication techniques at our disposal.
Eventually, we had a breakthrough. I had been exploring various linkage designs but Jon and I were concerned about the manufacturing costs and overall complexity of these models. Then I stumbled on discontinued toy, the Hoberman Flying Disc. It’s a simple expanding flying disc, like a Frisbee, but I discovered a few design tricks from it that could make the linkage approach feasible. I built a prototype inspired by, and in part made from, that disc. It had an expanding ring to accommodate different incision sizes, a membrane that delivered fluid, and suction at the bottom to prevent pooling and help remove incisional contamination.
When it was ready, I shared it with Insoo, the surgeon on our team. Insoo hardly ever liked anything, so when he smiled, I knew we had finally come up with something that could potentially to appeal to surgeons. I guess the lesson is to persevere and look for inspiration in unusual places!
 Jeremy Koehler, Insoo Suh and Jon Coe, co-founders of Prescient Surgical
Jeremy Koehler, Insoo Suh and Jon Coe, co-founders of Prescient Surgical
Reflecting on your experience, what advice do you have for other health technology innovators?
Jon: You can’t anticipate every challenge along the development path. Innovative approaches in translational medicine challenge ingrained habits and inherent biases. But, by the same token, innovators bring their own biases to the table. The key is to listen carefully to your potential users, iterate, and commit to a long-term partnership to solve their unmet needs.
Jon Coe, Jeremy Koehler, and Insoo Suh founded Prescient Surgical out of the Biodesign Innovation Fellowship in 2012. To learn more, visit the Prescient Surgical website.
Disclaimer of Endorsement: All references to specific products, companies, or services, including links to external sites, are for educational purposes only and do not constitute or imply an endorsement by the Byers Center for Biodesign or Stanford University.