Technologies
Niveus Medical Getting ICU Patients Back on Their Feet –
An Interview with Brian Fahey, founder of Niveus Medical
What is the need Niveus Medical set out to address?
Many critically-ill patients survive a prolonged stay in the intensive care unit [ICU]. But, by the time they’re discharged, some may have spent five or 10 days completely immobilized in bed – sometimes even longer. During that time, they develop full-body weakness that leaves them so debilitated that they usually can't walk, stand, or even sit up in bed on their own. The weakness they develop can require 6-12 months of rehabilitation, and often leads to significant morbidity and a dramatic impact on functional independence and quality of life. This is obviously a big deal for the patient, but it’s also a big deal for the hospital. Because of the way insurers pay hospitals for care provided in the ICU, the extended stays that are directly-attributable to ICU-acquired weakness can have tremendous negative financial impacts. Many critical care physicians are frustrated that that there’s not more they can do to prevent this type of ICU-related complication.
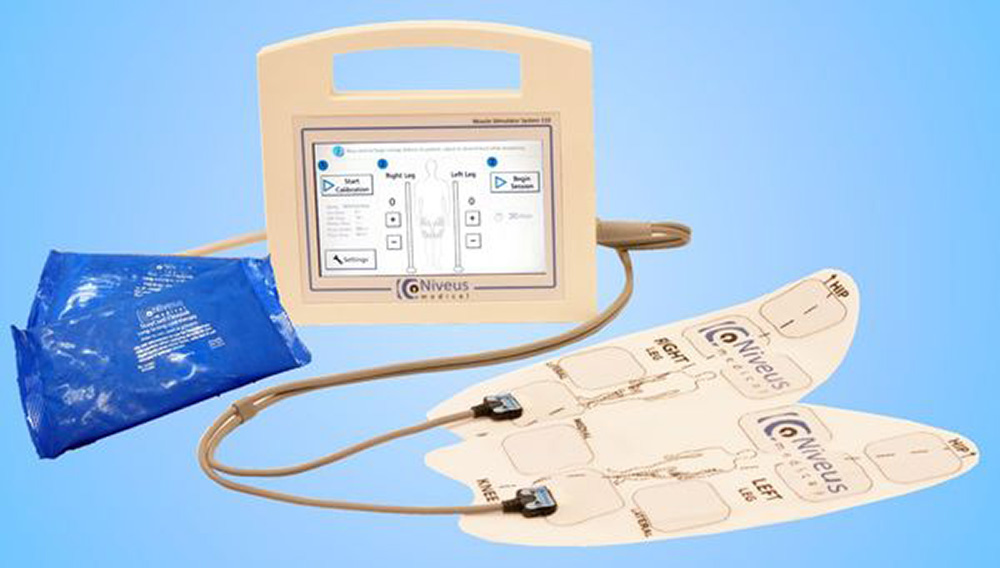
The Niveus Medical technologies were developed with the goal of helping patients preserve critical muscle strength. The muscle stimulation system provides ICU care teams with an adjunctive tool that can augment their care and allow them to be more proactive in addressing weakness.
What key insight was most important to guiding the design of your solution?
For a long time, the concept of ICU-acquired weakness was not widely understood. Once long-stay survivors left the ICU, the critical care medical teams didn’t have a reliable mechanism to track them and so oftentimes they didn’t know how they were doing. But as medicine evolved and treatments improved, there were more survivors and more clinical data to demonstrate how poorly many of these patients recovered. For example, a big New England Journal of Medicine study showed that more than 50% of survivors of acute respiratory distress syndrome weren't back to work at one year post-discharge.
“We wanted to intervene before patients lose their muscle mass in order to preserve the strength they have when they’re admitted, rather than allowing them to get weak and then trying to rebuild it.”
One existing and effective approach to combat the development of weakness in the ICU is early and progressive mobility. This type of intervention is great, but it can be really hard to do. And most hospitals aren’t great at mobilizing patients consistently. In many cases, you need three or four staff members to safely get these patients out of bed and walking. It can be complicated because the patients are connected to numerous tubes, wires, and lines, so there are significant fall risks. This motivated us to think about what can be done for patients while they're still in bed that that would require only limited involvement of ICU personnel and other resources – something that could augment any mobility efforts. We wanted to intervene before patients lose their muscle mass in order to preserve the strength they have when they’re admitted, rather than allowing them to get weak and then trying to rebuild it.
 Brian Fahey, founder of Niveus Medical.
Brian Fahey, founder of Niveus Medical.
How does your solution work?
Electrical muscle stimulation is used routinely as part of rehabilitation programs for a number of clinical conditions. There are numerous positive studies that demonstrate the clinical impact of muscle stimulation in both inpatient and outpatient settings. Clear benefits have been demonstrated in pulmonary, cardiovascular, surgical, orthopedic, and other cohorts of patients.
But a unique challenge associated with long-stay ICU survivors is that, in addition to systemic weakness, they typically develop a condition known as peripheral tissue edema [the accumulation of excess body water causing swelling]. They’re being pumped full of IV fluids for medical reasons and the body can't clear it all. All of that fluid accumulates in what's called the ‘third spaces.’ It's not in their vessels, it's not in their cells – they just swell like a sponge. And that makes it hard for a standard off-the-shelf muscle stimulator to work well. The extra layers of fluid impair the ability of muscle stimulators applied to the surface of the skin to deliver energy that reaches deep muscles.
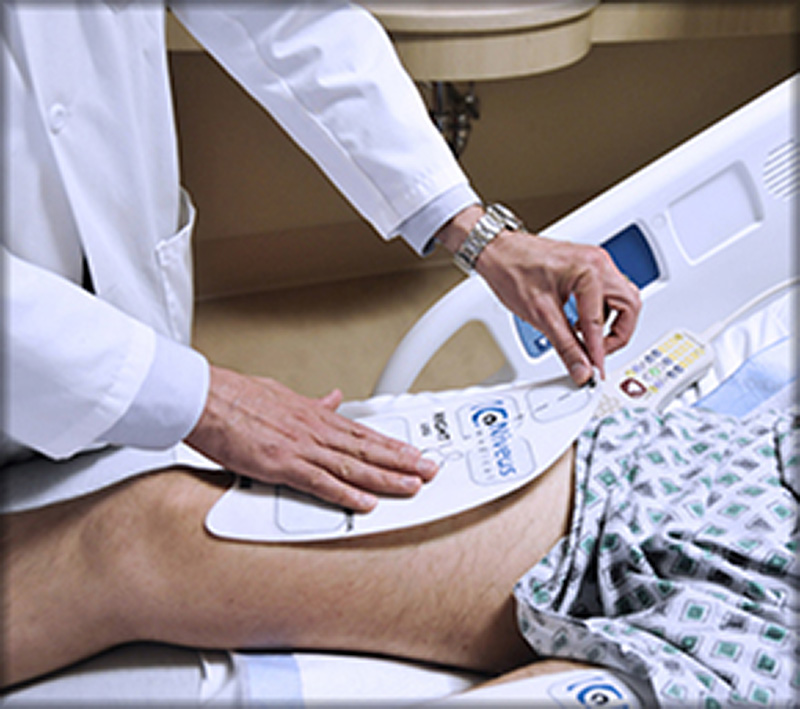
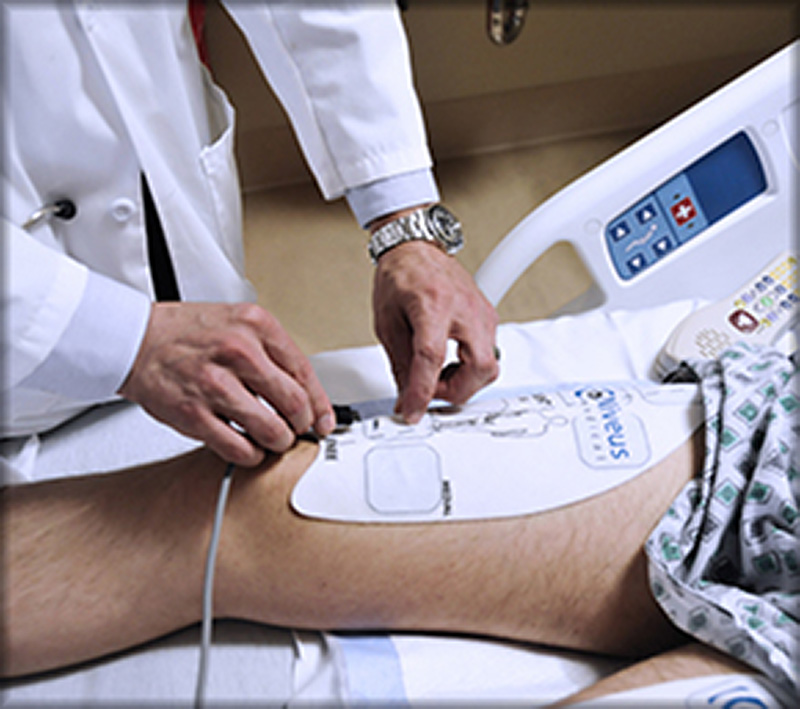
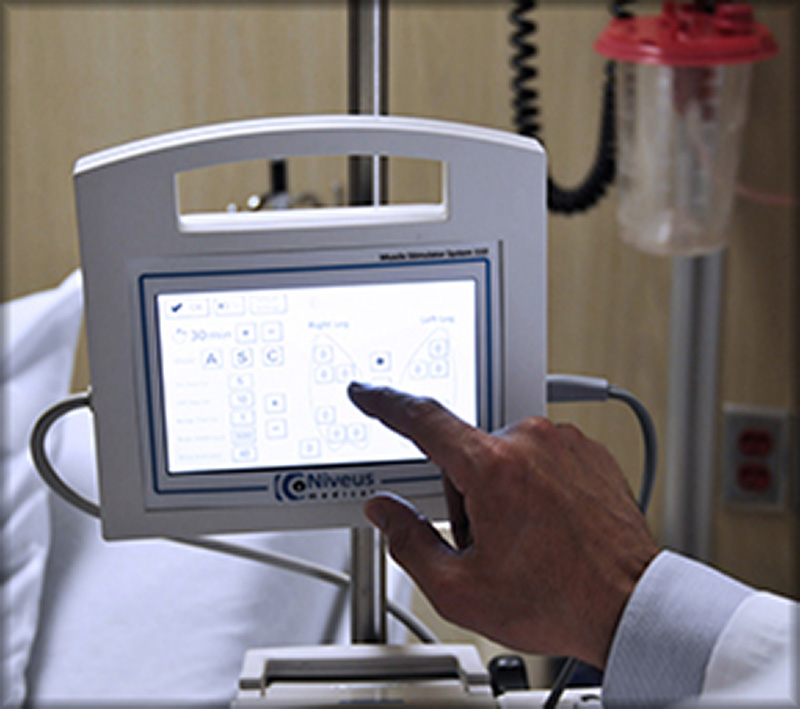
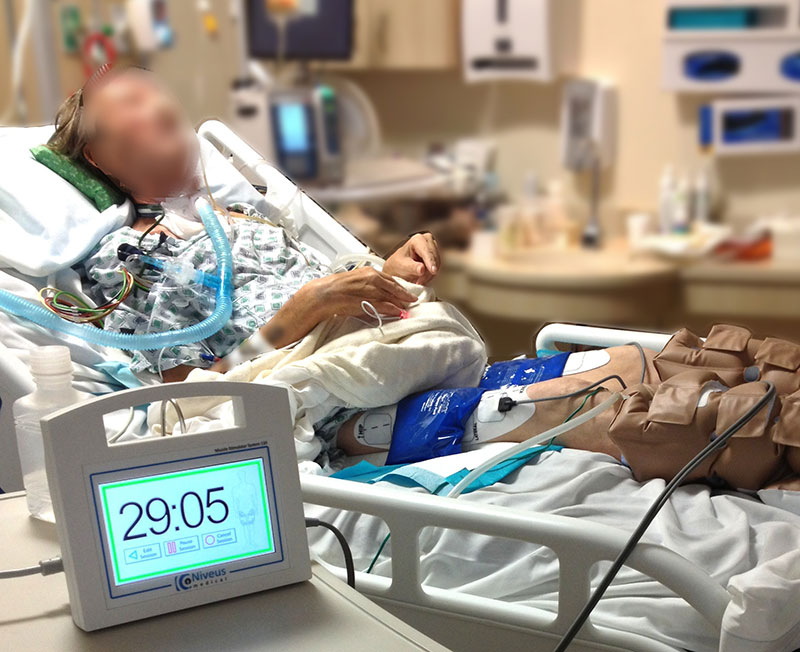 The Niveus Medical technology is intended to keep key muscle groups strong during bed rest and support faster patient recovery.
The Niveus Medical technology is intended to keep key muscle groups strong during bed rest and support faster patient recovery.
The Niveus solution addresses this challenge by combining thermal techniques with muscle stimulation. By combining local hypothermia with the muscle stimulation in a very specific way, it is believed that energy is driven deeper into the body to create stronger muscle contractions, which can generate better results.
At what stage of development is the solution?
The Niveus technologies helped patients during more than 10,000 treatments prior to being acquired by Stryker in 2017.
Tell us about a major obstacle you encountered and how you overcame it.
For us, technology development was relatively straightforward. Our greater challenge was finding the most appropriate way to prove the effectiveness of the technology in a manner that was reasonable for a modestly-funded start-up company. The ICU has rigorous expectations around evidence-based medicine. There aren’t a lot of therapeutic devices in the ICU; it’s mostly diagnostic equipment and drugs. The physicians are used to seeing new therapies backed by huge pharmaceutical trials.
As a small start-up, we could never afford to deliver trial results that included thousands of patients. But we had to clearly demonstrate that the technology worked. It took us a few iterations to get that right. In the beginning, we'd start at one hospital and prove to them that it worked through a series of internal evaluations. Then we’d take that data to the next hospital that was 10 minutes away and they’d dismiss the findings from the first hospital, saying things like, ‘Our patient mix is different, our protocols for treating patients are different.’ They didn’t view the results from another local hospital as applicable. So we’d have to begin a new set of evaluations to demonstrate to the second hospital that the technology was effective. The process was slow and tedious. But, ultimately, we were generally able to convince providers that using the technology on patients is beneficial and cost effective.
What role did your Biodesign training play in enabling you to design, develop, and/or implement this solution?
Biodesign taught me that understanding the needs of all relevant stakeholders is critical to success. It isn’t enough to understand the clinical problem or develop an interesting technology. You have to understand how your work fits into the big picture, especially since healthcare is such a dynamic and complex field. Biodesign didn’t necessarily give me those answers. But it taught me to ask the right questions and gather the right information to figure it out.
“You have to understand how your work fits into the big picture, especially since healthcare is such a dynamic and complex field.”
What advice do you have for other innovators about health technology innovation?
Be your own toughest critic. And be careful not to get blinded by your own passion. It’s important not to fall so in love with your own technology that you start to ignore or dismiss significant risks. The risks never go away, so it’s better to address them early.
Also, most medical technologies take 8-10 years to develop and commercialize. So most of us will only get four or five chances to start something in our careers. When you’re considering project, ask yourself whether you’re willing to wager 20% of your career – or more – on the idea. Your time is your most valuable asset. Make sure you spend it wisely.
Brian Fahey founded Niveus out of the Biodesign Innovation Fellowship in 2008. To learn more, visit the Niveus Medical website.
Disclaimer of Endorsement: All references to specific products, companies, or services, including links to external sites, are for educational purposes only and do not constitute or imply an endorsement by the Byers Center for Biodesign or Stanford University.