Technologies
iRhythm Diagnosing Suspected Arrhythmias:
An Interview with Uday Kumar of iRhythm
What is the need iRhythm seeks to address?
We wanted to find a better way to diagnose suspected heart rhythm problems, or arrhythmias, in non-hospitalized patients. The standard of care at the time was for the patient to wear a portable Holter monitor, a reusable Walkman-like device that has wires and electrodes you attach to the skin for 24-48 hours. But this solution didn’t work well for multiple reasons – it’s cumbersome to use, can’t be worn during showering or exercise, and isn’t kept in place long enough to detect many arrhythmias that occur briefly and intermittently. Additionally, the monitor has to be obtained and returned to the hospital, which can be a challenge for certain patient populations, in order for the physician to be able to access the diagnostic data. And typically only heart specialists prescribe these devices, so often a patient needs to be referred to a specialist first.
What key insight was most important to guiding the design of your solution?
There were several important insights that guided us. First, observing the cycle of care and how patients move through the system led to the insight that we needed a single-use product in order to get it into emergency rooms and primary care settings, which is where patients often go first when they’re experiencing symptoms. To get a device into an ER, for example, it has work like everything else in an ER – you open a packet and use it one time.
The second insight reflected the perspective of the patients. They needed discretion, comfort, and most importantly to get a diagnosis the first time rather than having to make repeat trips to the hospital, which is what often happened with the Holter monitor.
"[Patients] needed discretion, comfort, and most importantly to get a diagnosis the first time rather than having to make repeat trips to the hospital..."
The third important realization was around what diagnostic information we needed and how to present it to different physician stakeholders. The data had to cover a diagnostic period of 1-2 weeks. For non-specialists, we needed to provide a clear top-line finding -- whether the patient had an arrhythmia or not -- that was immediately actionable in terms of referral to a specialist for treatment. But we also had to make sure more comprehensive data were available for heart specialists who needed to dig deeper to guide their treatment. Without that, specialists would often order repeat testing, which was time-consuming and costly.
 Members of the early iRhythm team, including Uday Kumar (second from left).
Members of the early iRhythm team, including Uday Kumar (second from left).
Along these lines, the fourth insight was that it was imperative to make the entire solution cost-effective for payers. In this case, it meant not developing the highest tech solution possible. For example wireless connectivity and real-time data transmission were starting to take-off when we were developing our solution. Had we incorporated these capabilities into our device, it would have been too expensive and too inaccessible for the majority of patients. Instead, we kept it simple so that the greatest number of patients possible could benefit from the solution.
How does your solution work?
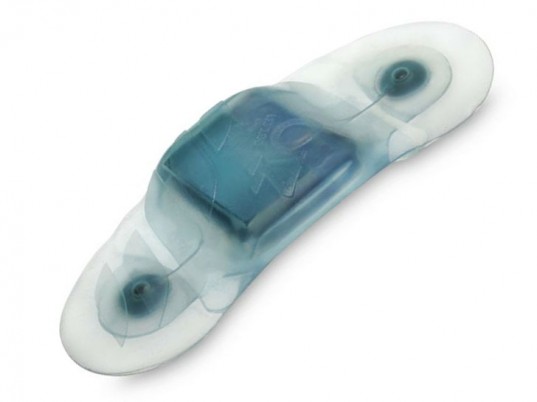
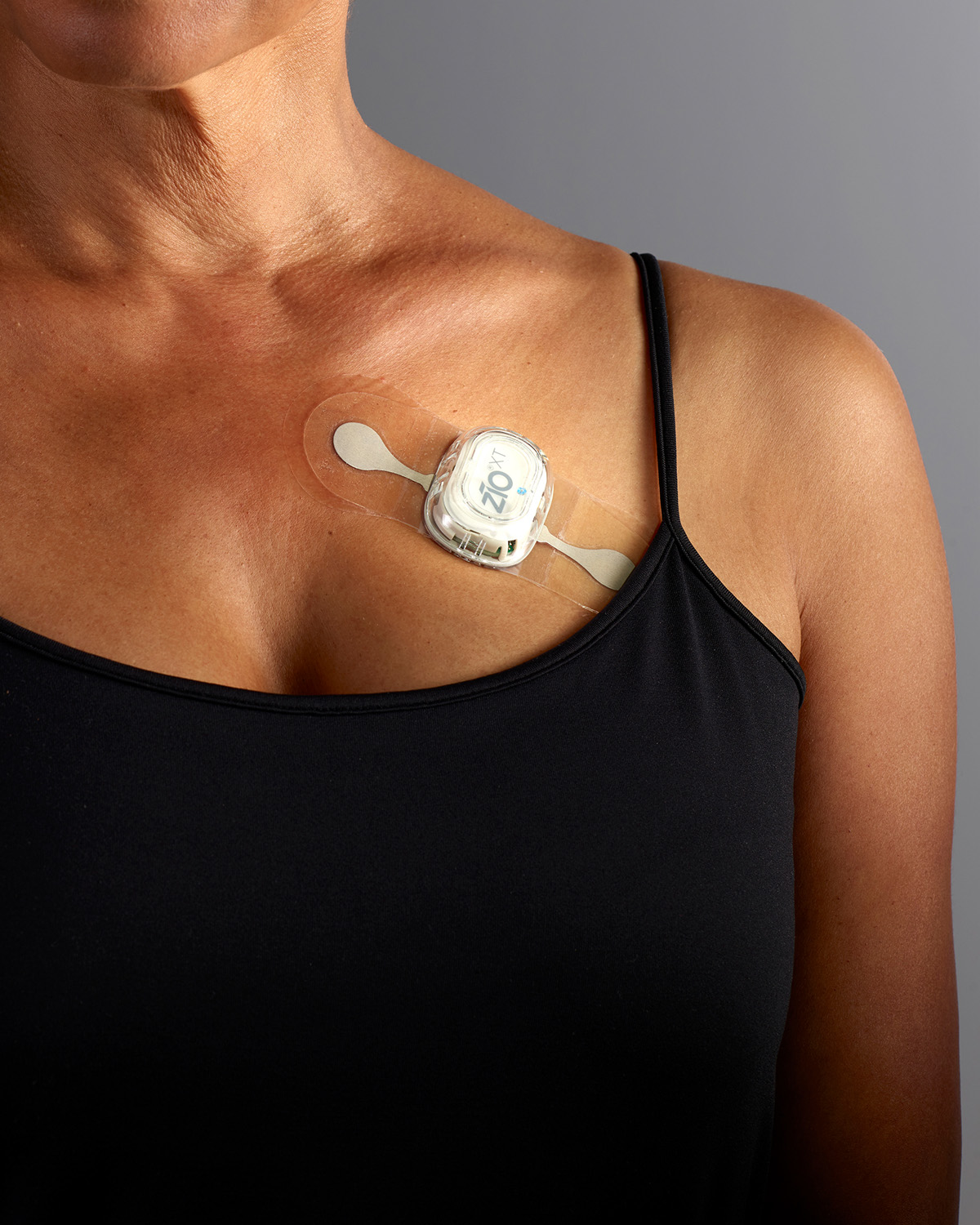
The Zio Patch is a small, flexible, water-resistant, adhesive monitoring patch that records heart rhythms continuously for up to two weeks. The solution is inexpensive and simple enough that it can be given to patients of all ages and abilities, not only by heart specialists, but also in the emergency room or at a primary care physician’s office. At the end of two weeks, the patient mails the patch to an iRhythm Zio service center via the postal service. There, the data is downloaded and a diagnostic report is generated.
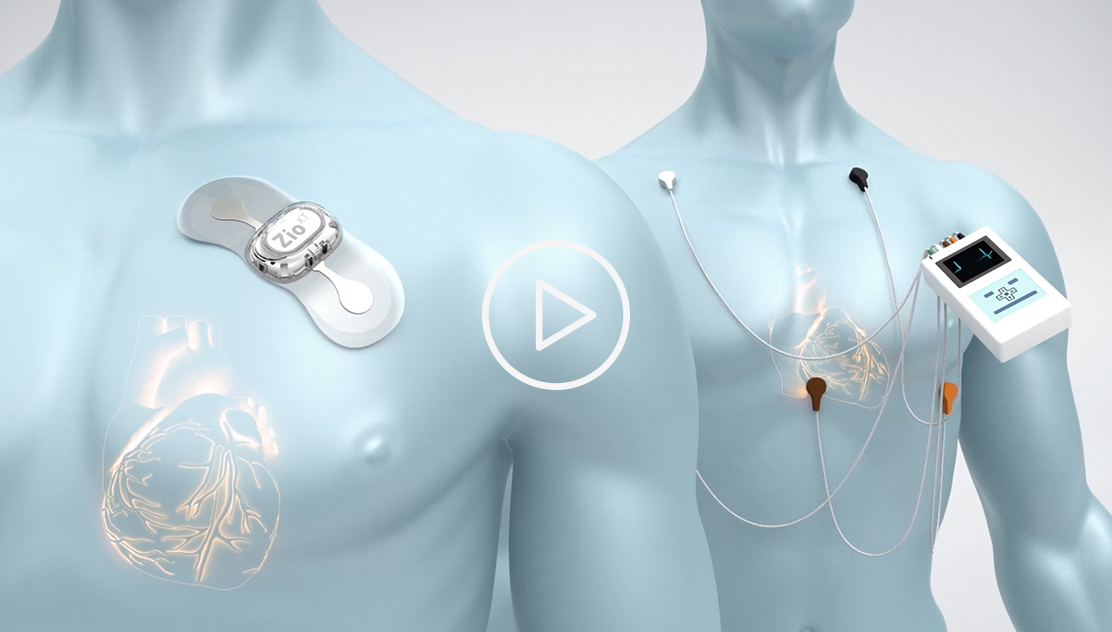 This short promotional video, provided by iRhythm, illustrates how the company's system is intended to work (see endorsement disclaimer below).
This short promotional video, provided by iRhythm, illustrates how the company's system is intended to work (see endorsement disclaimer below).
What’s important is that the doctor who prescribes the device and the doctor who interprets the report don’t have to be the same – the patient can get the patch wherever they are, but have the data interpreted by a heart specialist who decides whether a specialist visit is necessary. So we’re moving the testing to the patient in the primary care office or ER and moving the information to the specialist who can then readily determine a treatment plan.
At what stage of development is the solution?
The Zio Patch and its associated algorithm were cleared by the FDA in 2009, and the technology is now widely available.
What are your plans for the future?
I’m no longer personally involved in the day-to-day operations of iRhythm, but the company has recently started to market a new product that’s targeted at helping high-risk patients who can benefit from the inclusion of real-time data transmission. Over time, they may also start looking at other disease areas where this paradigm of collecting diagnostic information and delivering it to the right stakeholders in an efficient manner can be helpful to even more patients.
Tell us about a major obstacle you encountered and how you overcame it.
Reimbursement. It wasn’t insurmountable, but we didn’t anticipate how long it would take to get there and how much money would be needed. Early on, when we talked to physicians, they saw that our solution was not only better for patients, but more efficient in terms of being able to obtain an accurate diagnosis. But we had to work with each insurer, individually, to try to make that case.
Fortunately, we were lucky to get support and early adoption from the Veteran’s Administration and integrated healthcare systems like Kaiser, which recognized the value of the solution up front. As an integrated system, they pay for both the primary care and specialist visits. So it was intuitive for them to want to monitor a patient first in order to determine whether a specialist visit was actually needed, rather than sending them to the more costly specialist for the initial diagnosis. Over time, we leveraged our experience with those systems to demonstrate our value to early insurer adopters. Then, as we accumulated more data and published more articles on clinical outcomes, it became easier to get more insurers on board.
"As we accumulated more data and published more articles on clinical outcomes, it became easier to get more insurers on board."
How did your Biodesign training help you advance the project?
The Biodesign approach was instrumental to the iRhythm project. Specifically, it provided a framework for breaking down the complex systems problem of how patients get monitored into smaller ones, each with its own need criteria. For instance, the data collector, or patch, had to be simple and not too complex for patients but our data processing, a cloud-based algorithm, had to be very technologically sophisticated to provide comprehensive information and allow efficient scaling. Applying the Biodesign process to understand these needs separately allowed us to create the best overall solution. Since iRhythm I’ve used the process to launch multiple companies and I’m even more confident that it’s the most effective way to do health technology innovation.
Uday N. Kumar founded iRhythm out of the Biodesign Innovation Fellowship in 2006. To learn more, visit the iRhythm website.
Disclaimer of Endorsement: All references to specific products, companies, or services, including links to external sites, are for educational purposes only and do not constitute or imply an endorsement by the Byers Center for Biodesign or Stanford University.