Technologies
Ciel Medical Preventing Ventilator-Associated Pneumonia:
An Interview with Kate Garrett and Dan Azagury of Ciel Medical
As Biodesign Innovation Fellows, what is the need you set out to address?
Kate: When critical care patients can’t breathe on their own, they are put on a ventilator for support. However, the ventilator itself can cause complications, most significantly ventilator-associated pneumonia, or VAP. VAP is one of the most commonly acquired infections in the ICU and also one of the most life-threatening.
It’s also expensive; it’s estimated that VAP adds $40,000 to the hospital bill, which has put this complication under the scrutiny of the Centers for Medicare and Medicaid Services (CMS). Although they haven’t yet declined to reimburse for VAP, hospitals are acutely aware of the need to reduce VAP from both a quality-of-care and cost perspective.
“Ventilator-associated pneumonia is one of the most commonly acquired infections in the ICU and also one of the most life-threatening.”
Dan: The problem is that the endotracheal tube itself can serve as a conduit for bacteria, or act as a reservoir for pathogens by providing a place for oral secretions to pool and potentially be aspirated into the lungs. The tube prevents the patient from coughing to dislodge the fluids, and suctioning them is difficult because they can’t be reached by standard oral suctioning methods. Our goal was to find a way to prevent aspiration in long-term ventilated patients to reduce the risk of pneumonia.
What key insight was most important in guiding the design of your solution?
Kate: By the time we were fellows in 2011, breathing tubes with built-in suction lines had been on the market for 10 years. There was strong clinical evidence that this technology significantly reduced the risk of VAP by facilitating the removal of pneumonia-causing fluid from the airway. But the adoption wasn’t there – most hospitals were using these specialized tubes infrequently if at all.
Dan: We were baffled until we started to watch intubation procedures in the ICU. It’s a high-stress, high stakes procedure and there’s no time for the caregiver to ponder different breathing tube options. The tubes with built-in suction lines are more expensive – so they’re not the standard of care – and there’s no way for the physician doing the procedure to know which patients will need the suction lines at the time of intubation.
“Intubation is a high-stress, high-stakes procedure. There’s no time to ponder different breathing tube options.”
Kate: So this sparked an idea for us – what if we could find a way to deliver suction post-intubation? That way the physician could choose to add suction later if there appeared to be a risk of VAP. This led to our original need statement: “A more cost effective way to achieve subglottic suctioning for ventilated patients in the ICU regardless of endotracheal tube type.”
How does your solution work?
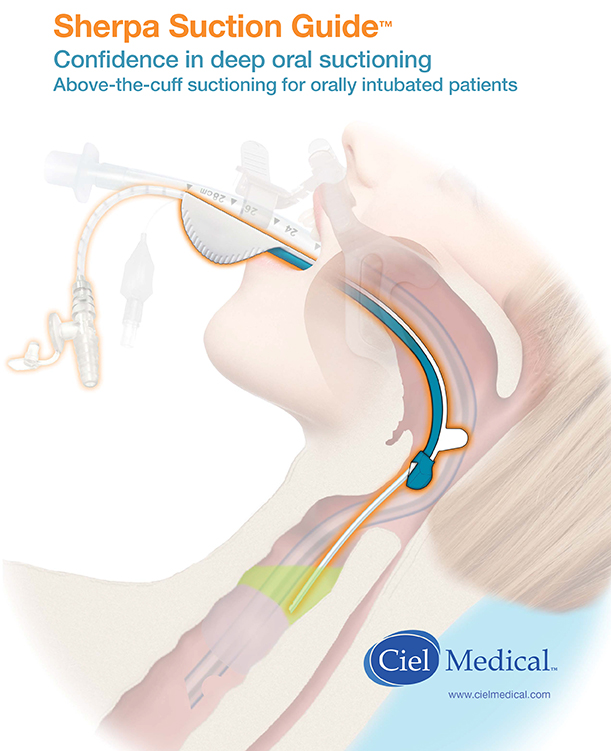
Dan: We developed a delivery guide that enables subglottic placement of an independent suction catheter. The delivery guide clips to the underside of the endotracheal tube and uses the tube as a guidewire as it is advanced through the back of the mouth. A suction catheter is then passed through the guide and advanced to the cuff of the endotracheal tube, and pooled secretions are suctioned out using a manual control. The guide is then removed, and the catheter is closed and left in place clipped to the endotracheal tube for future use.
At what stage of development is the solution?
Kate: The Sherpa Suction System met FDA requirements in 2015. The first year of commercialization was focused on working closely with early clinical partners to perfect the design and training materials required for successful adoption. After developing a second product that facilitates accurate placement of endotracheal tubes and a third product for securing catheter lines to endotracheal tubes, Ciel Medical was acquired by Vyaire Medical in 2017. Vyaire Medical launched a commercial roll-out of the Sherpa Suction System across their 50+ sales reps in the United States in late 2017.
Tell us about a major obstacle you encountered and how you overcame it
Dan: One of the biggest challenges we had to overcome involved designing the delivery device for the catheter so that we could get the catheter to the right place.
Kate: Previously, caregivers would try to deliver the suction catheter blindly. You can't see where you’re going because of the curvature of the airway. The opening to the trachea is pretty small and it's on the anterior side of the body, so you're trying to thread the needle, and it’s nearly impossible. The guide makes the process much easier, but it wasn't simple to design because the variation in curvature is really significant from patient and patient, and so is the length. There's also a lot of anatomy that we had to be able to get past, and our device had to track over a wide range of endotracheal tube sizes.
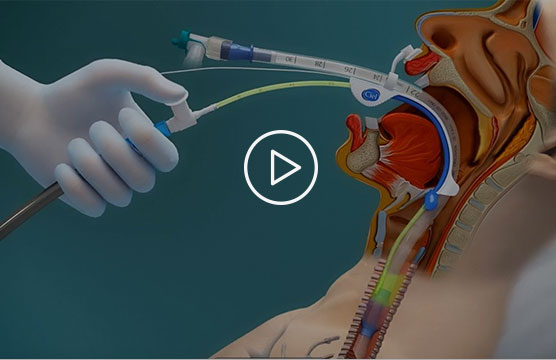 This short promotional video created by Ciel Medical illustrates how the Sherpa Suction Guide technology is intended to work (see endorsement disclaimer below).
This short promotional video created by Ciel Medical illustrates how the Sherpa Suction Guide technology is intended to work (see endorsement disclaimer below).
Dan: Getting to the final design seemed like an endless process. There wasn’t a single key insight that opened the door for us, we just kept trying and gaining in sophistication. We developed literally hundreds of prototypes. We did a lot of work on cadavers, and each time we were going into the cadaver lab, Kate would bring in with another bag of like eight different variations; some with very minor permutations and some more significant. And we'd go in and try them all and see.
Kate: The key was trying to get the right mix of ease-of-use and controllability. For example, in our first model, we instructed caregivers inserting the guide to stop at the teeth. It’s a clear visual indicator, but we found that people would advance the guide too far anyway. So we realized that we had to make it so that you could not advance the guide any farther than the teeth. There was also a lot of variation in the form factor at the tip of the guide. We had dozens of different iterations.
Dan: Another challenge that came up through initial use cases was that people tend to bite the device when you put it in their mouths. We hadn’t anticipated this. If a patient bites at some point as you’re pushing through, it will leave a dent in this plastic. And because the space is very tight, that tiny dent means you can’t push the catheter in and out anymore, and you're stuck.
Kate: But if we made it more rigid so that people couldn't dent it by biting, we would lose the flexibility we needed for navigating. So we ended up making little slits on the top so that it is still flexible and accommodates the curvature we need, but has a rigid bottom.
Dan: The opinion leaders in the field loved it. It was the first time they’d seen something that would actually work from a physical perspective and a hospital economic perspective too.
 Dan Azagury and Kate Garrett, co-founders of Ciel Medical
Dan Azagury and Kate Garrett, co-founders of Ciel Medical
Reflecting on your experience, what advice do you have for other health technology innovators?
Kate: Make sure to take assumptions and technology solution ideas out at the beginning, and really focus one the root problem. Take the time to really understand the problem from the perspective of the users and patients. When you think about it that way, you are automatically thinking about the flow of money and the value proposition in the hospital. How somebody going to pay for this? Where can I save them money? What's the benefit from an overall cost perspective?
Kate Garrett and Dan Azagury founded Ciel Medical out of the Biodesign Innovation Fellowship in 2012.
Disclaimer of Endorsement: All references to specific products, companies, or services, including links to external sites, are for educational purposes only and do not constitute or imply an endorsement by the Byers Center for Biodesign or Stanford University.